MENU
The SARS-CoV-2 Variant Detection Kit is intended for molecular test of H69/V70 wild/deletion unique to United Kingdom (UK), and E484K unique to South Africa (SA)/Brazil (BZ) of SARS-CoV-2 S gene (Spike protein) in upper respiratory specimens such as Nasopharyngeal swab, Oropharyngeal swab.
| RADI COVID-19 Detection Kit | |
|---|---|
| Intended use |
Qualitative detection of SARS-CoV-2 variants (UK, South Africa/Brazil) |
| Specimen type | Previously extracted RNA for COVID-19 positive samples |
| Target |
UK wild : H69/V70 UK variant : H69/V70 deletion South Africa/Brazil variant : E484K IPC : Human RNase P |
| Product contents |
Primer & Probe Mixture (Brown) 3X RT MasterMix (Yellow) Positive Control (Red) RNase free water (Blue) IFU |
| Storage condition |
-25 ℃ ~ -15 ℃ |
| SHELF LIFE | 6 months |
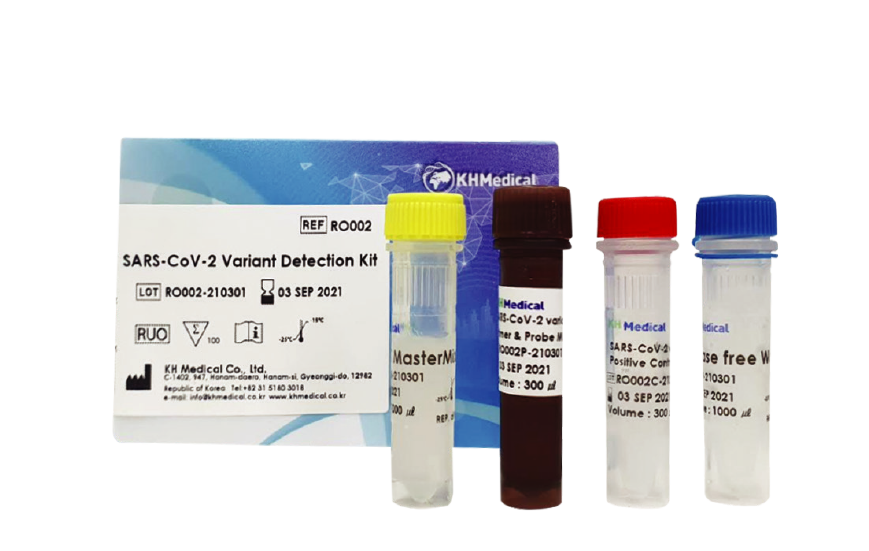
No sequencing required for detection of S gene variants
| Variant of Concern | Lineage | Mutation |
|---|---|---|
| UK | B.1.1.7 | H69 / V70 deletion |
| South Africa | B.1.351 | E484K |
| Brazil | P1 | E484K |

* Simplified Procedure when make mixture
(Number of tubes minimized)!

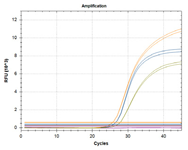
| Cases | Targets | Result | |||
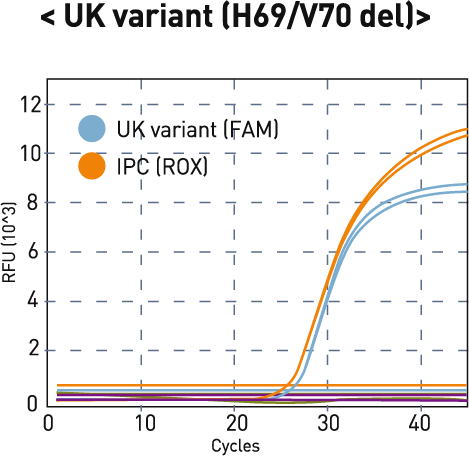
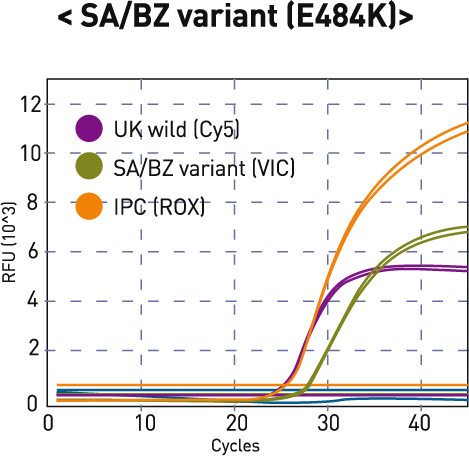
| UK | SA/BZ | IPC* (ROX) |
|||
| H69/V70 wild (ROX) |
H69/V70 del (FAM) |
E484K (VIC) | |||
| 1 | + | - | - | + | Wild (No variant) |
| 2 | - | + | - | + | UK variant (H69/V70 del) |
| 3 | + | - | + | + | SA/BZ variant(E484K) |
| 4 | - | + | + | + | UK, SA/BZ variant (H69/V70 del, E484K) |
| 5 | - | - | - | - | Invalid (Re-testing) |
| CATEGORY | PRODUCT NAME | DESCRIPTION | CAT. NO. |
| PCR Kit | SARS-CoV-2 Variant Detection Kit | 100 tests/Kit | RO002 |
| CATEGORY | PRODUCT NAME | DESCRIPTION | CAT. NO. |
| PCR Kit | RADI COVID-19 Detection Kit | 100 tests/Kit | RV008 |
| Manual Extraction Kit | RADI Prep swab and stool DNA/RNA Kit | 100 tests/Kit | MP002 |
| Automated Extractor | RADI PREP Plus | upto 8 samples | KM001 |
| RADI Extractor 32 | upto 32 samples | OE005 | |
| RADI Extractor 192 | upto 192 samples | OE006 | |
| PCR instrument | RADI PCR 96 | 96 samples | OE004 |
| Sample collection Kit | RADI Transport Medium Kit | 50, 100 | OD103 |